Q. Here’s a question from Twitter: Can you explain to me what the M protein in multiple myeloma is?
A. Multiple myeloma is a malignant, clonal disorder of plasma cells that originates in the bone marrow. It’s a relatively common disorder, accounting for 1% of all malignancies and 10% of all hematologic malignancies in adults. Patients present with painful, lytic lesions of the bones, recurrent and persistent infections, weakness, renal failure, and hypercalcemia. The prognosis is generally not great, but new chemotherapeutic agents seem to hold some promise.
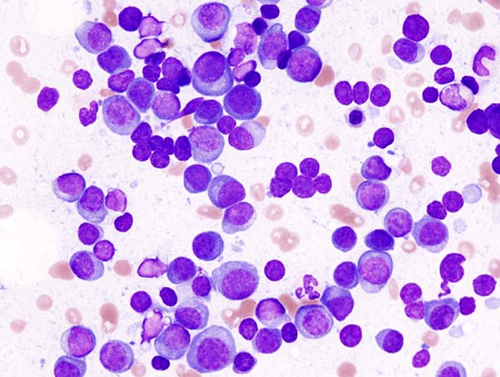
Patients with myeloma have a monoclonal proliferation of plasma cells in the bone marrow, meaning that there are a ton of malignant plasma cells that all originated from the same initial cell. In the bone marrow aspirate above, you can see tons of malignant plasma cells. A few look a lot like plasma cells, with clock-face chromatin and a hof and everything, but others look for all the world like blasts. That’s one thing to remember about myeloma – malignant plasma cells don’t always resemble their nice little benign counterparts.
The malignant plasma cells almost always secrete immunoglobulin, and because they are monoclonal, they all secrete exactly the same form of immunoglobulin. This is very different than what you see in a patient without myeloma, where there are a bazillion different types of plasma cells, all making different types of immunoglobulin molecules. This huge mass of all-the-same immunoglobulin secreted by myeloma cells is called a “monoclonal gammopathy.” (Normal immunoglobulins are called “polyclonal.”)
Monoclonal gammopathy is so characteristic of myeloma that you can use it for both diagnosis of disease and follow-up of patients. You can detect the monoclonal immunoglobulin using serum electrophoresis (which separates the blood proteins into groups based on charge and size). There’s a predictable pattern of proteins in normal serum: albumin, the most abundant protein in the blood, migrates to a certain predictable point; other proteins migrate to different places, which are given different names: the alpha 1 region, the alpha 2 region and the beta region. Immunoglobulins migrate to a unique place called the gamma region, and because normal immunoglobulins are all slightly different, they migrate to slightly different places within that region, giving a gentle bell-shaped curve or smear (depending on whether you’re looking at a tracing or the actual bands on the gel).
In myeloma, the immunoglobulin is monoclonal, so it all migrates to exactly the same spot on the gel! Which gives you a big spike (if you’re looking at a tracing) or a very distinct, crisp, strong band (if you’re looking at the gel itself). This spike is called an M-spike (you could remember M for either monoclonal or myeloma), and the corresponding monoclonal protein that it represents is called an M protein.
A few other things to note about this M protein:
1. You need to do electrophoresis on urine too, not just serum. Some cases of myeloma secrete only light chains (these are called Bence-Jones proteins), which are so small that they are passed in the urine. So if you only did a serum electrophoresis, you’d miss them.
2. While patients with myeloma have an increase in the total amount of immunoglobulin present in the blood (due to the large monoclonal immunoglobulin spike), they also have a decreased amount of normal, polyclonal immunoglobulins. So when you look at an electrophoresis, you’ll see this huge spike in the gamma region, but also a noticeable depression in the amount of the background normal immunoglobulins.
3. A little trivia regarding the kinds of immunoglobulin expressed by myeloma cells. The most common heavy chain expressed in myeloma is IgG (60%); next is IgA (20%). Rare cases express IgD or IgE, and IgM myeloma is virtually nonexistent (most cases of plasma cell lesions that express IgM turn out to be Waldenström macroglobulinemia). Almost one-fifth of all cases of myeloma secrete only light chains. And somewhere between 1 and 5% of all cases of myeloma secrete no detectable immunoglobulin at all! Which, without the familiar M-spike, would make for a pretty difficult diagnosis.







Thank you Kristine. Lebon
That you Kristine!
Clear, concise explanation…
Thanks, helped alot.
Sub epithelial deposits along with m spikes are seen in which renal condition…..
If M spike is only in Serum test but Not in Urine Test, what does it mean?
It means that the monoclonal immunoglobulin is in a form that cannot be excreted by the kidney (which is the case for most immunoglobulins!). Light chains, by the way, are easily excreted in the urine (they’re called Bence-Jones proteins) – so if you have a myeloma that just secretes light chains, you’d have a monoclonal immunoglobulin in the urine, but not in the blood. This is one of the main reasons we always order both a serum and a urine electrophoresis.
I have been taking a small amount of frankincense each day for the last six months, an essential oil. My m-spike went from 1.2 six months ago to .6 last week.
that really helped in touching the basics
thanks a lot!
b blessed
Hi,
I just received a call from my doctor who on a routine blood test found that I have M spikes of 0.08. He has suggested I see an oncologist immediately. I have an appointment on December 1st.
I’ve been reading all these sites, and I now realize that although having an M spike of any number is abnormal, does it always indicate a cancerous or dangerous disease. Are there any other reasons for having it?
I have had a torn rotator cuff for a year now, and the second surgeon has decided it should be operated on. As I will be in a sling for a month, I’ve decided to wait until Spring. I was wondering if having an inflammation in ones system for a year, as I have with my torn rotator cuff, could it cause this type of response on my blood test?
If one has an M spike does it always mean cancer? Please advise. Thank you.
Lee
Hi Lee –
No – an M spike does NOT always mean cancer. Many people develop something called monoclonal gammopathy of undetermined significance (MGUS) which is a benign condition in which you have a small M spike. This occurs with greater frequency in older patients. Sometimes MGUS develops into myeloma (or another malignant disease) but most of the time it does not. Having a large M spike (over 3) does tend to go along with malignancy – but yours is so low that I doubt it indicates myeloma. It would still be a good idea to have it investigated just to be sure – and then to follow the spike to make sure it doesn’t go up. In answer to your second question, no – having a lot of inflammation would not lead to an M spike. Let me know if you have other questions!
Hi,
I have been suffering from Multiple Myeloma for the last two and a half years. my M band was 5gm/dL and after taking chemo(Bartezomib) for six months the M band disappeared completely and it remained like that for around a year. now again this time, when the SPEP was done M band is raising its head, as the reports show 200mg/dL M band. Does it mean that Myeloma is coming back? If yes when should I go for chemo again? and shall I be given the same salt i.e. Bartezomib or some other salt has come into the market? I am a general surgeon living in India.I request you to kindly guide me.
thanks in anticipation.
Hi Dr. Modi –
I wish I could give you a good answer but your question would be better directed to someone who practices clinical hematology. I know about the pathology part – but the clinical treatment of myeloma, like many hematologic diseases, is constantly changing – and it would be important to get an opinion from someone who practices in the field. Best of luck!
Kristine
I Apologize if ths gets printed twice not sure first time went,i recently got blood test back shows 0.4 M spike, Iron saturation low 14 and Immoglobin e 136 . I have a follow up with Hem. in 2 weeks but would like to prepare my self scared to death I may have multiple mel. My daughter a nurse said my spike is very low. Wondering if I will need treatment etc.?
Hi Linda –
Your M spike is indeed very low. Usually such a low spike is not of concern for myeloma (in myeloma, the magnitude is usually above 3.0 g/dL). I would try not to worry (easier said than done, I know!) – and see what the hematologist recommends. He/she will likely simply want to follow you with periodic checks to make sure it isn’t creeping up. Many people develop a monoclonal spike as they get older – and when it is so small, it usually is not ominous at all. The iron saturation will need to be investigated too – most likely that is a totally separate issue. You’d want to also get a serum iron and a ferritin level to sort out the different types of anemia that have iron abnormalities (iron-deficiency anemia and anemia of chronic disease are the two main ones). Best of luck! Let us know how it goes 🙂
I donate at Biolife Plasma and they have detected an M spike. (really SMALL) that has always been the same since donating for over 3 years. Now it’s an issue. Anything I should be worried about? The nurse said just a tiny edge up in my curve. ???
Hi Mary – I would not worry too much about it. Many people develop a small M spike as they age – and most of the time it doesn’t turn into anything dangerous. You may wish to see a doctor to be evaluated and to get exact numbers for the M spike. Most likely they will just want to follow you periodically to make sure it doesn’t rise precipitously. Best of luck!
Wow how informative! My MSPIKE over the last 4 months is 0.90.70.8…Had a bone marrow, 20 bone x-rays, and Catscan. My Onclo/Hemo Dr. said not to worry with these numbers. All is good but to have blood tests in 6 months to see if it elevates. Like most I was worried to dealth since I’m a 11 year squamious cell carcinoma survivor ( Base of Tonque)But be positive have faith and you’ll be fine …Rich Daytona Beach
Excellent info….
my m band is 0.37g/dl…. have multiple lytic lesions in vertberas and a a pathological fracture in C5 which I am going to be operated… seeing an oncolgist tonmmorow
bone marrow biopsy still not done
all other blood parameters are well within range inclining CBC calcium etc….
I am a doctor myself and double guessing by reading informative articles as yours
Thank you, Kristine. Your words have put me at ease even though I plan to follow up with periodic blood work every 4-6 months to check my M-spike level. I was just told today for the first time that I got a 0.1 result. I am going on 51 and hope this is just a sign of getting older. Thanks again for the information.
Excellent!
This is awesome information nice easy to understand definition of MM symptoms and treatments. I wish this postings, would remain or repeat at the top of my Myeloma group so that the same questions arnt repeated every day.
Can you have 2 different m-spike number?
You could, but it would be a very rare case! M stands for monoclonal (or sometimes myeloma) – which means that there is a group of cells that is secreting the exact same type of immunoglobulin. There is such a thing as a biclonal plasma cell malignancy (two clones of cells, each secreting their own type of immunoglobulin) but that is very uncommon.
You are WONDERFUL. Thank you for the info
Very helpful. Thank you.
I just read this site for the first time and I am so impressed with Kristine’s clearly and carefully written definition and explanations re multiple myeloma. For me and my husband, it’s always been difficult to grasp what this disease is all about.
I have too many questions to ask right now- it’s very late and time to go to bed, but I will be here again. Thank you.
Welcome, Isabel! I hope you can find some good information here 🙂
I can’t tell you how grateful I am that I came across this site! I’ve just started the process of an oncologist and the testing for myeloma. Your answers are so direct and understandable – wish I could take you to my appointments!
Thank you for all you do!
Hello Kristine,
My numbers are 4% and .3g/dl, not .03 as I reported it the second time. I thought I should clarify that.
By the way the work of a pathologist sounds fascinating. At the same time it is so much responsibility. Not everyone could tolerate knowing that someone else’s health depended on your expertise.
Thank you again.
Lilac
Hello, I just came across this site. I found everyone’s comments and info very helpful. I wish August/2016 I would have had this info and been better prepared. I went to a Dr. For , not this but severe sweating. The Dr. Called me end of August and said, ” referring you to Oncologist “. I asked her, ” do you think I have Cancer”? She didn’t know, she said. I was being referred and I would become their patient. I was so terrified. I still don’t have a diagnosis from that practice.i was told by Oncologist that it was MGUS and I had all the testing and Oct. 1st he said you don’t have C. I see him April 1st. For follow up. This is the first time I’ve gotten back on computer since this all started. I’m going to write down some questions for this visit.. I cried so much and so hard both times I saw him that I couldn’t even speak let alone ask a question. He must think I’m a nut. Except to leave the house for test, I sat on the sofa for two months and googled and cried. I am crying now just thinking and writing about all that went down those months. .. when I asked the Dr. That day when she called and said she was referring me and I didn’t need to see her again.. what does that tell you? I finally said. I came to you for sweating and now I’m seeing an oncologist. She said it’s all related.. it is not related.. the referring Dr. Told me I had an M protein months later when I saw him. He said this entire situation was handled poorly..
I thank you so much for your info. I wish you great success in your career. I just wish someone had tried to explain it to me like you did or at least tried, maybe I would have heard something through all the tears!!
Bless you. Sorry I was so long in this post
Thank you so much for this site! I was diagnosed 11 years ago and for the first time I know what an M Spike is. Woohoo. Maybe a stupid question, but how are the numbers arrived at? Do people actually count them, or computers do the counting? Thanks
Not a stupid question! There are lab tests that measure the amount of protein; for blood, it’s reported in grams of protein per deciliter of blood.
My mspike is 0.3 I’m scared have cancer that is a possibility
Hi Yanet –
I can understand your fear! When you hear “M spike” the big fear is that it’s myeloma.
However, in myeloma, the M spike needs to be a big one (like 3 grams per deciliter of blood).
It sounds like your M spike is small (I’m assuming that when you say 0.3, that you mean 0.3 g/dL). That’s nowhere near the amount of monoclonal immunoglobulin (or M spike) you see in myeloma.
It is not uncommon to find small amounts of monoclonal immunoglobulin in patients without myeloma – particularly as we get older.
I am a pathologist, however – and I would advise you to discuss this result with the physician who ordered it for you. He or she will explain what this small M spike means, and discuss what type of follow-up is best.
I hope that helps!